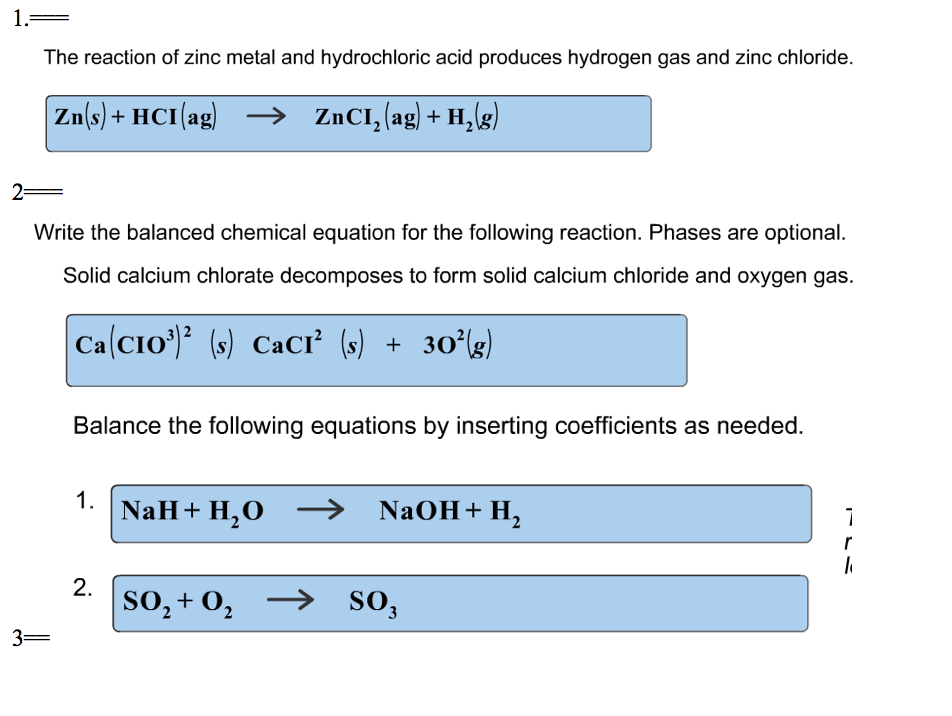
To complete their octet stability criteria Ag being a metal donates its 1 electron to. As a general rule, if you balance the molecular equation properly, the net ionic equation will end up being balanced by both mass and charge. AgCl lewis structure is an ionic compound made up of ions Ag+ and Cl. In the above equation, the overall charge is zero, or neutral, on both sides of the equation. Balancing by charge means making sure that the overall charge is the same on both sides of the equation. Balancing by mass means ensuring that there are equal masses of each element on the product and reactant sides. Net ionic equations must be balanced by both mass and charge. However, it is not absolutely necessary to order the reactants in this way. charge density during anodic formation of AgCl on a Ag wire in. 
This is somewhat customary because that is the order in which the ions must be written in the silver chloride product. Keywords: Ag/AgCl electrode, chloride sensor, anodization, open circuit. it is demonstrated that: (1) embrittlement in chloride environments can. Notice that in writing the net ionic equation, the positively-charged silver cation was written first on the reactant side, followed by the negatively-charged chloride anion. high negative charge, e.g., AgCl4 3, Ag(SCN)4 3, and Ag(S2O3)3 5, the. NH 3 ( aq) + HCl ( aq) NH 4+ ( aq) + Cl - ( aq) These complexes dont react with hydrochloric acid, even at 100 o C. Silver halides are ionic crystals consisting of a regular cubic lattice of Ag and halide ions together with a small proportion of defects, such as Ag ions that have been displaced from their regular lattice position to another interstitial position (the Ag ions are much smaller than the halide ions), and the corresponding vacancy in the lattice. In order to test the supposition of AgCl monolayer formation, the charge density contained in peaks P and Pc for a Ag wire electrode was measured and found to. The net ionic equation is the chemical equation that shows only those elements, compounds, and ions that are directly involved in the chemical reaction. By itself, ammonia reacts rapidly with hydrochloric acid to form ammonium chloride.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
